The Mechanism of a Battery

Batteries are a familiar and essential source of energy in our lives. A battery uses a chemical reaction to generate electricity and outputs that electricity externally. This exhibit explains the mechanism of batteries and the different types of batteries.

Use your own body to become a battery!
If you put your hands on two different metal panels, you become a battery and electric current flows. Do different combinations of metals cause any changes?
For more details, check the exhibit panels.
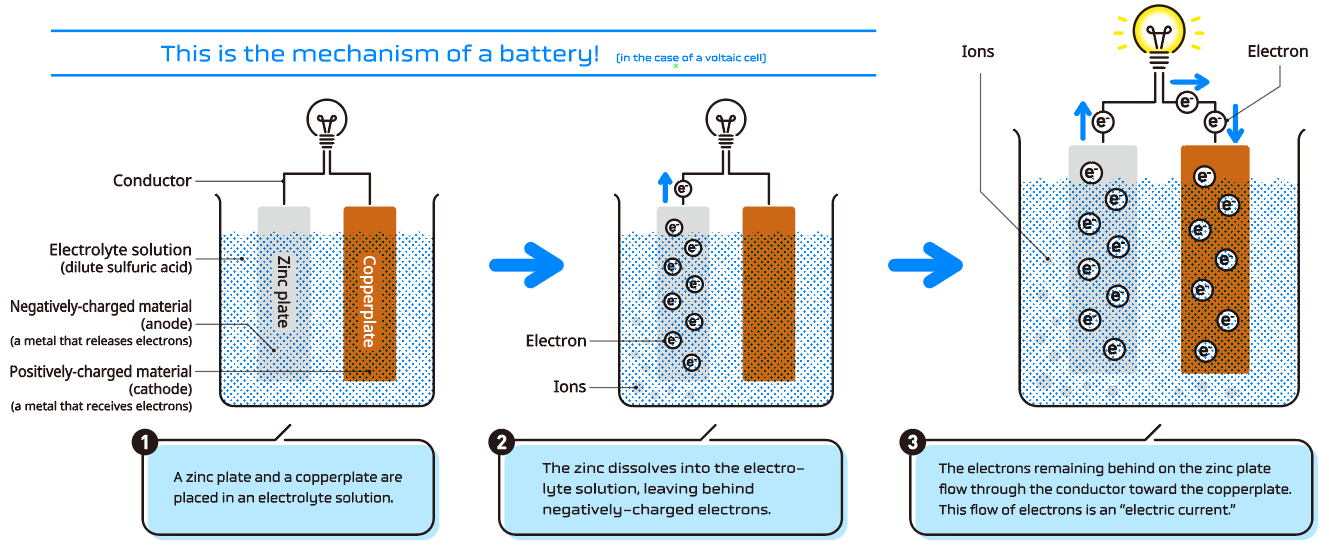
When positive and negative electrodes made from two different types of metals are immersed in an electrolyte solution and connected, metal ions from the negative electrode dissolve into the electrolyte solution and electrons move through the conductor to the positive electrode. Electricity is generated from this reaction.
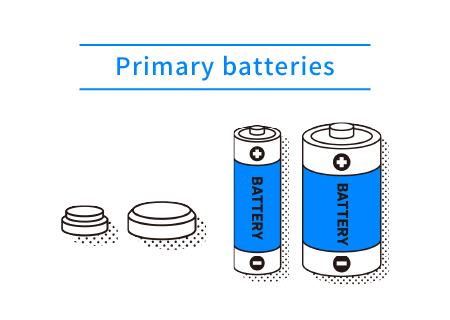
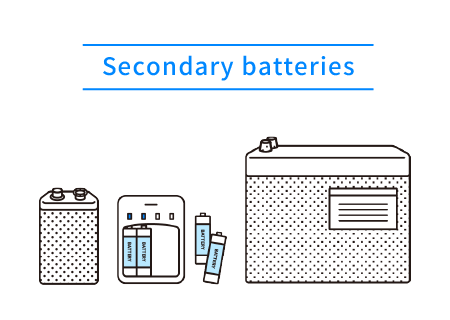
There are two types of batteries: batteries that cannot be charged, known as primary batteries, and those that can be charged, known as secondary batteries. The main types of primary batteries are manganese and alkaline batteries, and the main types of secondary batteries are lithium-ion and nickel-metal hydride batteries.